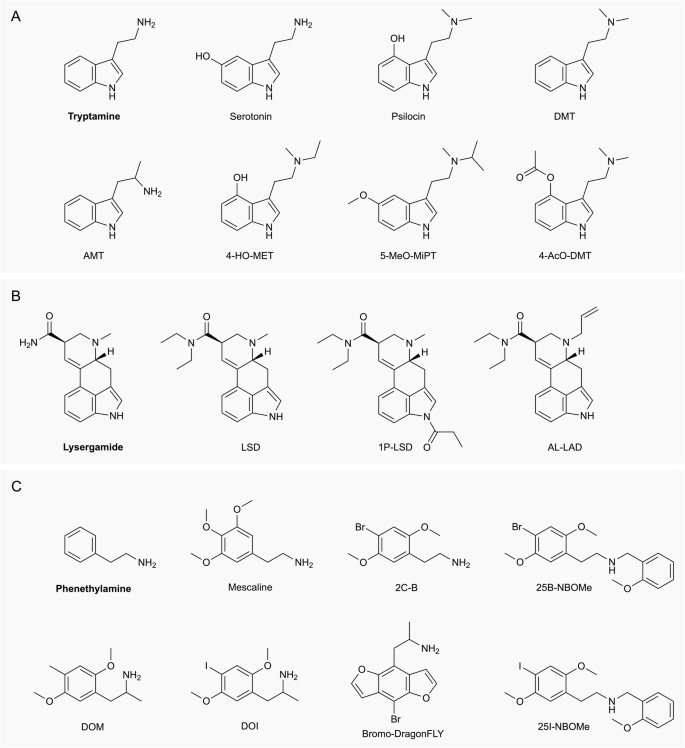
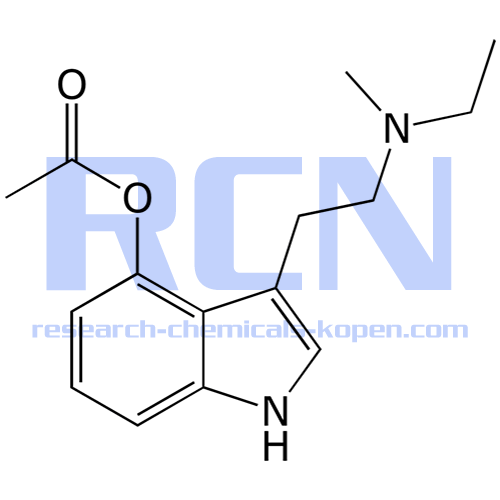
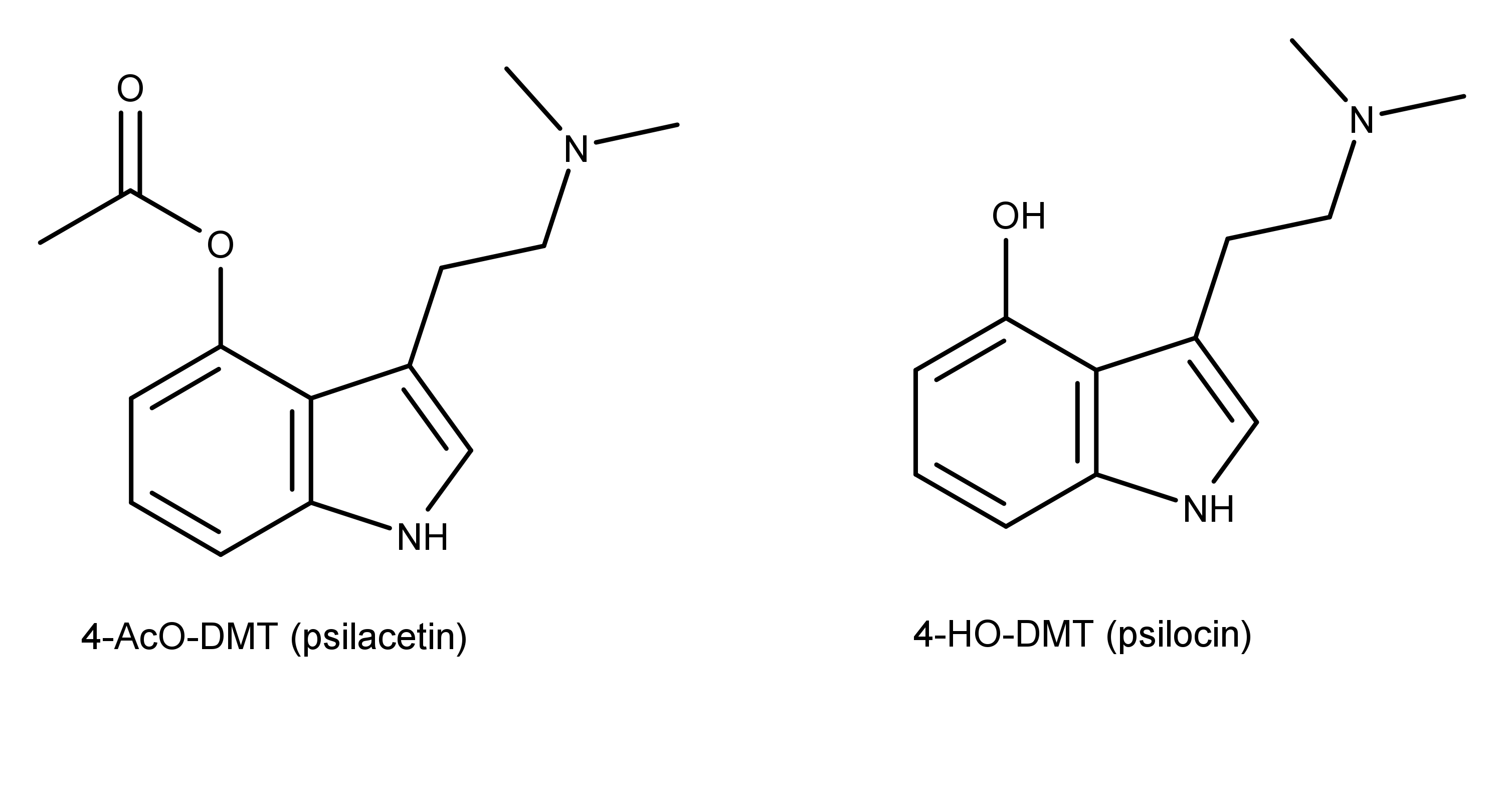
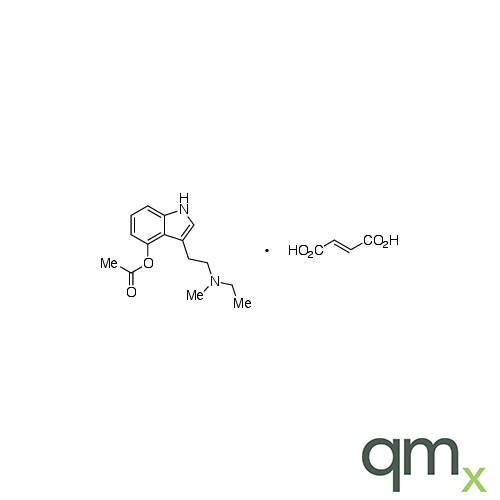
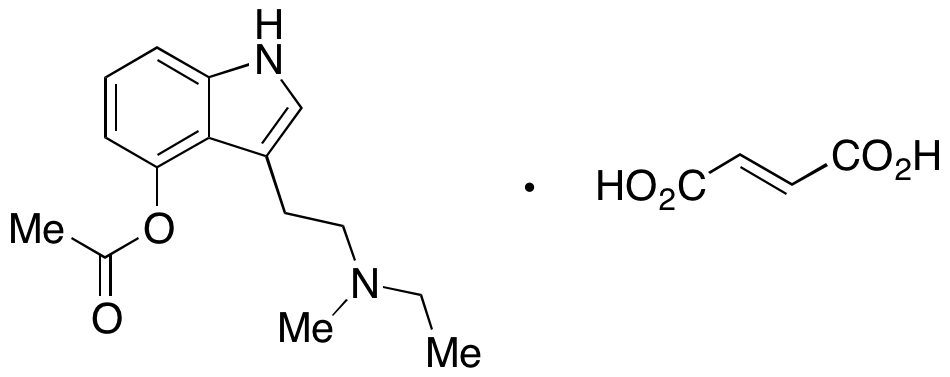
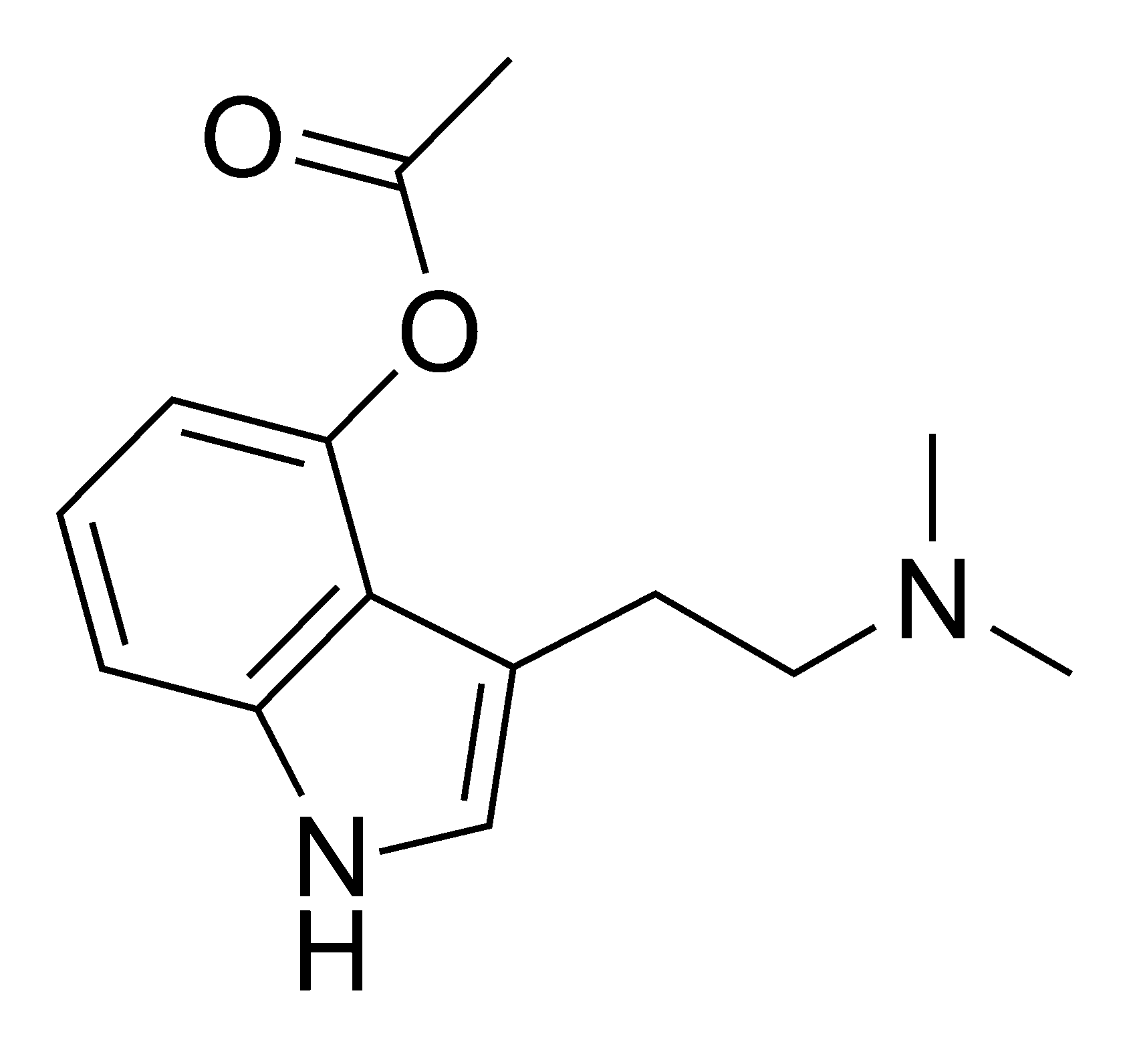
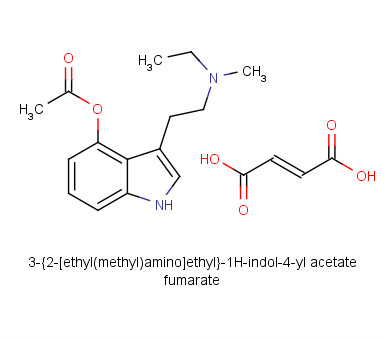
4-Acetoxy-N-methyl-N-ethyltryptamine Fumarate (4-AcO-MET Fumarate; 4-Acetoxy -MET Fumarate) 1.0 mg/ml in Dimethyl Sulfoxide (as free base)

Novel psychoactive substances: overdose of 3-fluorophenmetrazine (3-FPM) and etizolam in a 33-year-old man | BMJ Case Reports

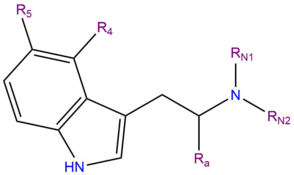
Receptor Binding Profiles for Tryptamine Psychedelics and Effects of 4-Propionoxy-N,N-dimethyltryptamine in Mice | ACS Pharmacology & Translational Science
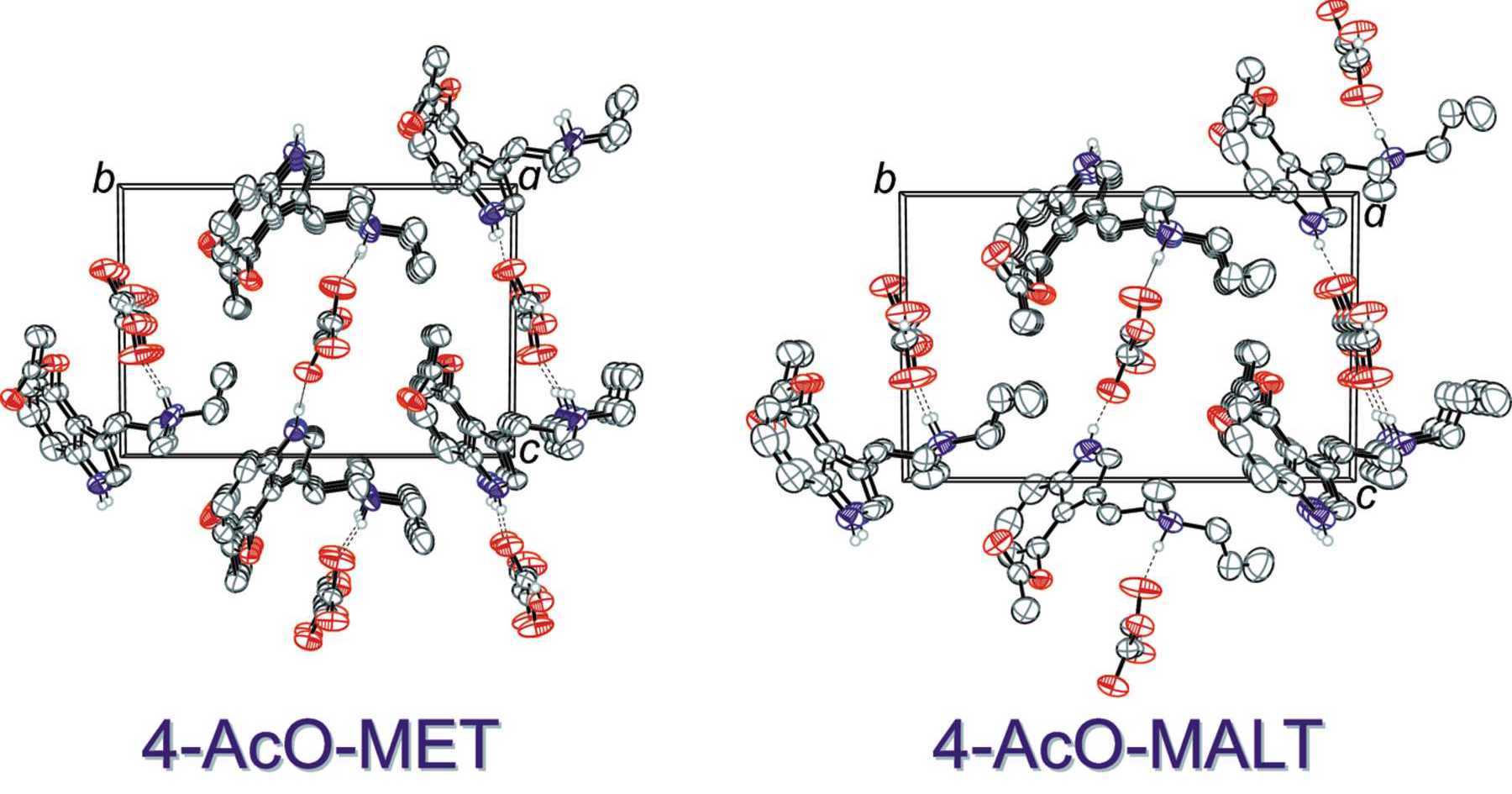
Psilacetin derivatives: fumarate salts of the meth­yl–ethyl, meth­yl–allyl and diallyl var

4-acetoxy DET (hydrochloride) (4-acetoxy-N,N-Diethyltryptamine, Ethacetin, Ethylacybin, CAS Number: 2749504-15-0) | Cayman Chemical

Active Metabolite of Aeruginascin (4-Hydroxy-N,N,N-trimethyltryptamine): Synthesis, Structure, and Serotonergic Binding Affinity | ACS Omega
Psilacetin derivatives: fumarate salts of the meth­yl–ethyl, meth­yl–allyl and diallyl var

Structure–Activity Relationships for Psilocybin, Baeocystin, Aeruginascin, and Related Analogues to Produce Pharmacological Effects in Mice | ACS Pharmacology & Translational Science

4-acetoxy DMT (fumarate) (4-acetoxy-N,N-Dimethyltryptamine, O-Acetylpsilocin, Psilacetin, CAS Number: 1217230-42-6) | Cayman Chemical